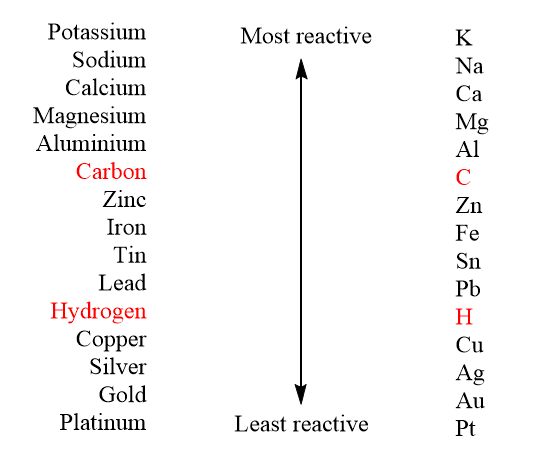
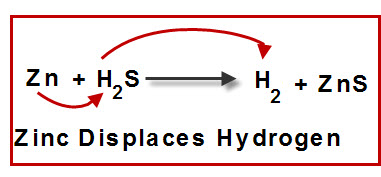
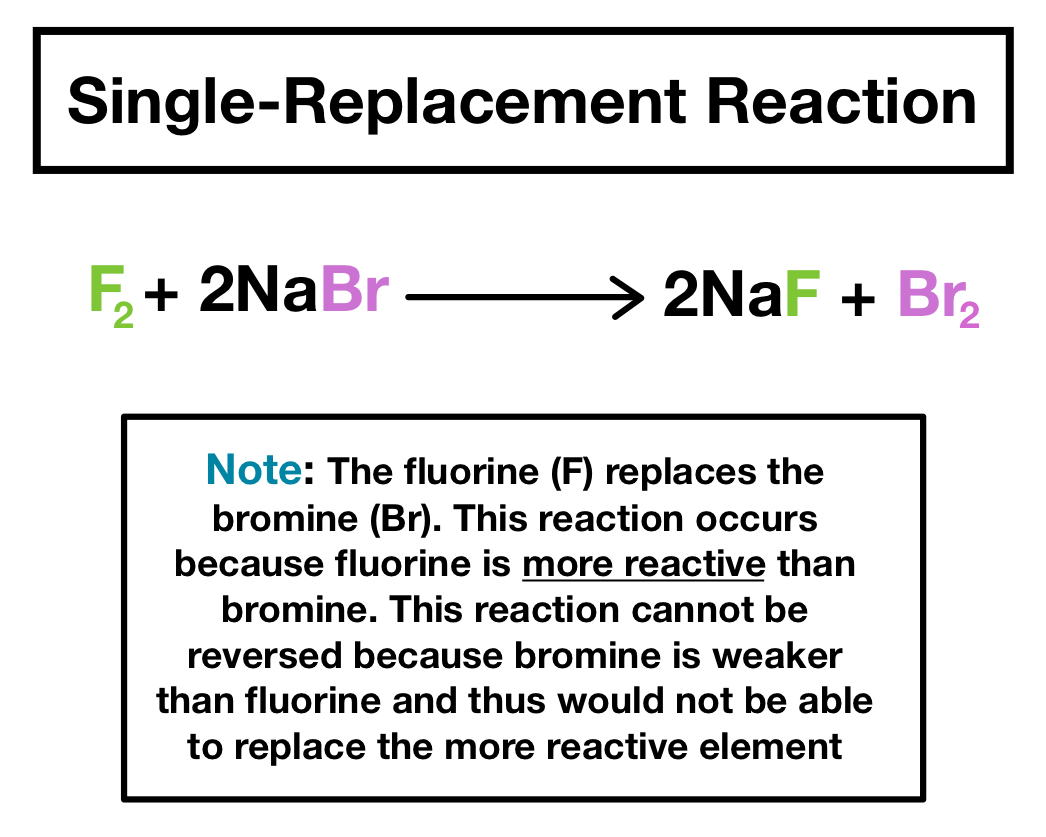
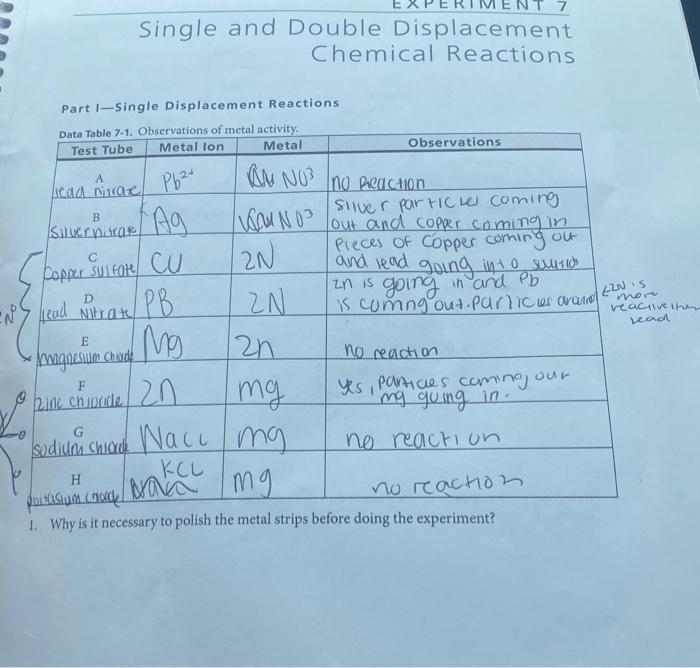
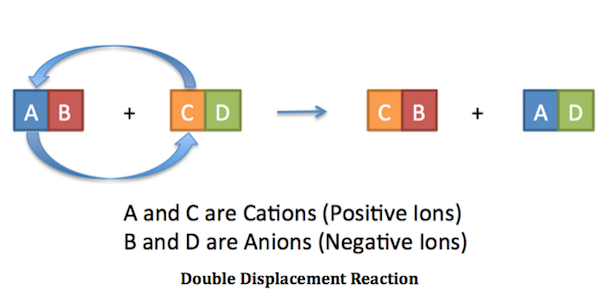
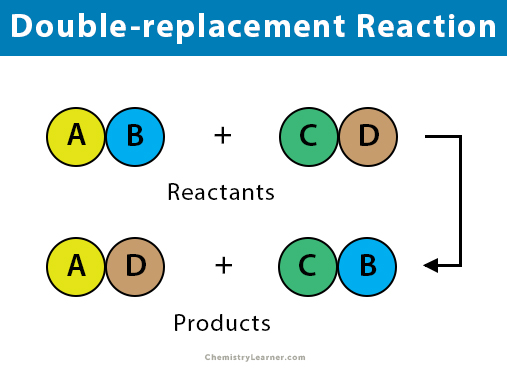
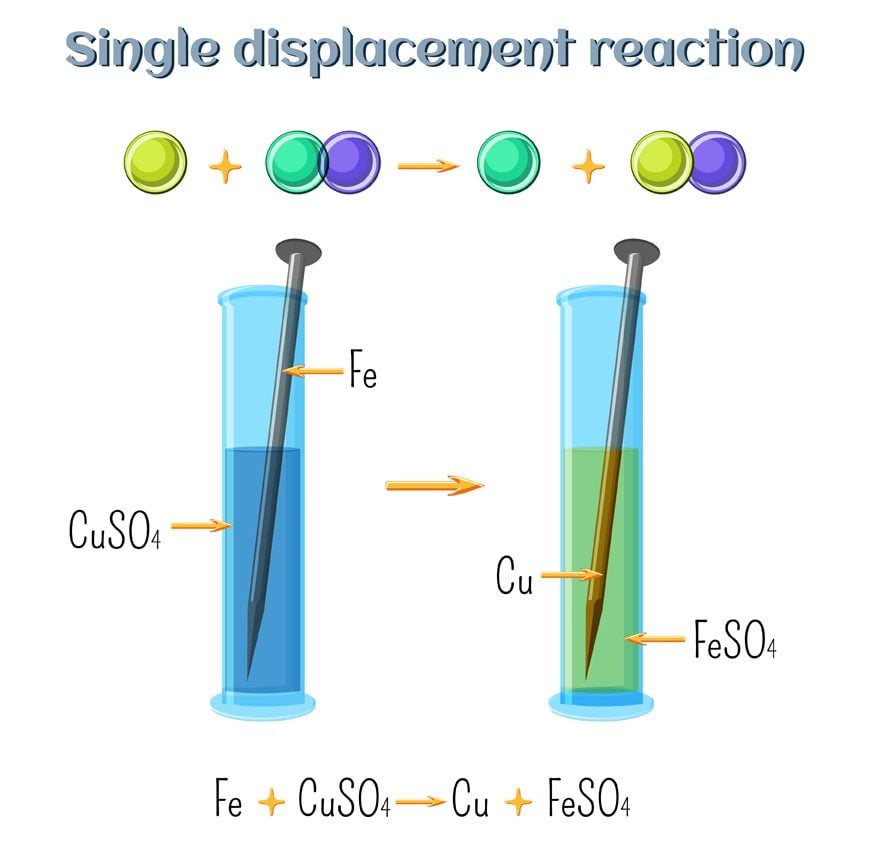
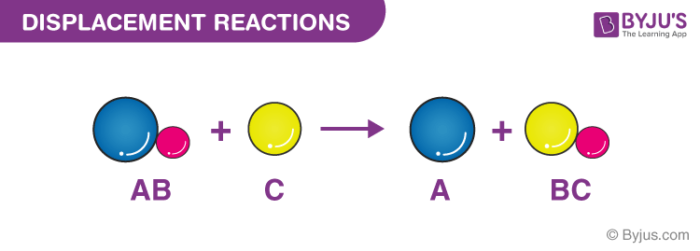
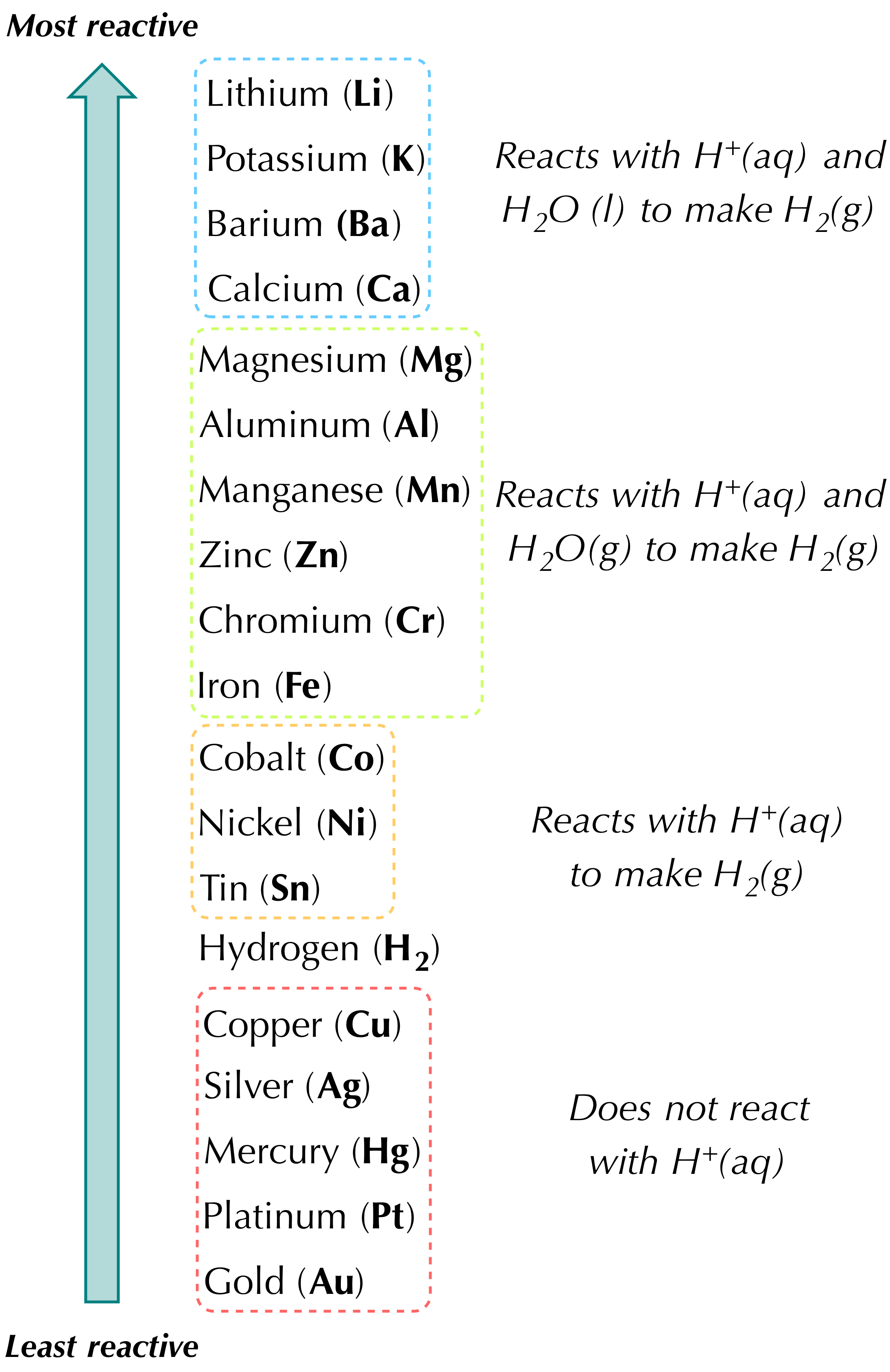
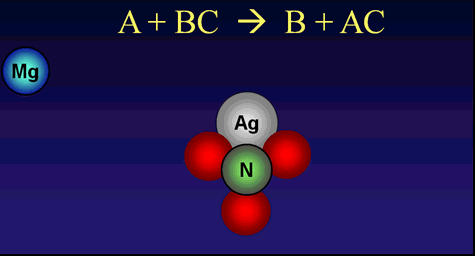Reaction Types Chemistry Dr. May Single Displacement Reaction A Compound and an element form a compound and an element A Compound and an element form. - ppt download
What are the differences between single and double replacement reactions? What are their similarities? - Quora